|
This two-cohort comparison study included andexanet alfa patients enrolled at US hospitals from 4/2015 to 3/2020 in the prospective, single-arm ANNEXA-4 study and a synthetic control arm of 4F-PCC patients admitted within a US healthcare system from 12/2016 to 8/2020. We evaluated the effectiveness and safety of andexanet alfa versus 4F-PCC for management of apixaban- or rivaroxaban-associated intracranial hemorrhage (ICH). 
Four-factor prothrombin complex concentrates (4F-PCC) are commonly used as an off-label, non-specific, factor replacement approach to manage FXa inhibitor-associated life-threatening bleeding. alfa is approved (FDA "accelerated approval" EMA "conditional approval") as the first specific reversal agent for factor Xa (FXa) inhibitor-associated uncontrolled or life-threatening bleeding. 
8 Evidence-Based Practice Center, Hartford Hospital, Hartford, CT, USA. 7 Department of Pharmacy Practice, University of Connecticut School of Pharmacy, 69 North Eagleville Road, Unit 3092, Storrs, CT, 06269, USA.6 Department of Global Health Economics and Outcomes Research, Alexion, AstraZeneca Rare Disease, 121 Seaport Blvd, Boston, MA, 02210, USA. 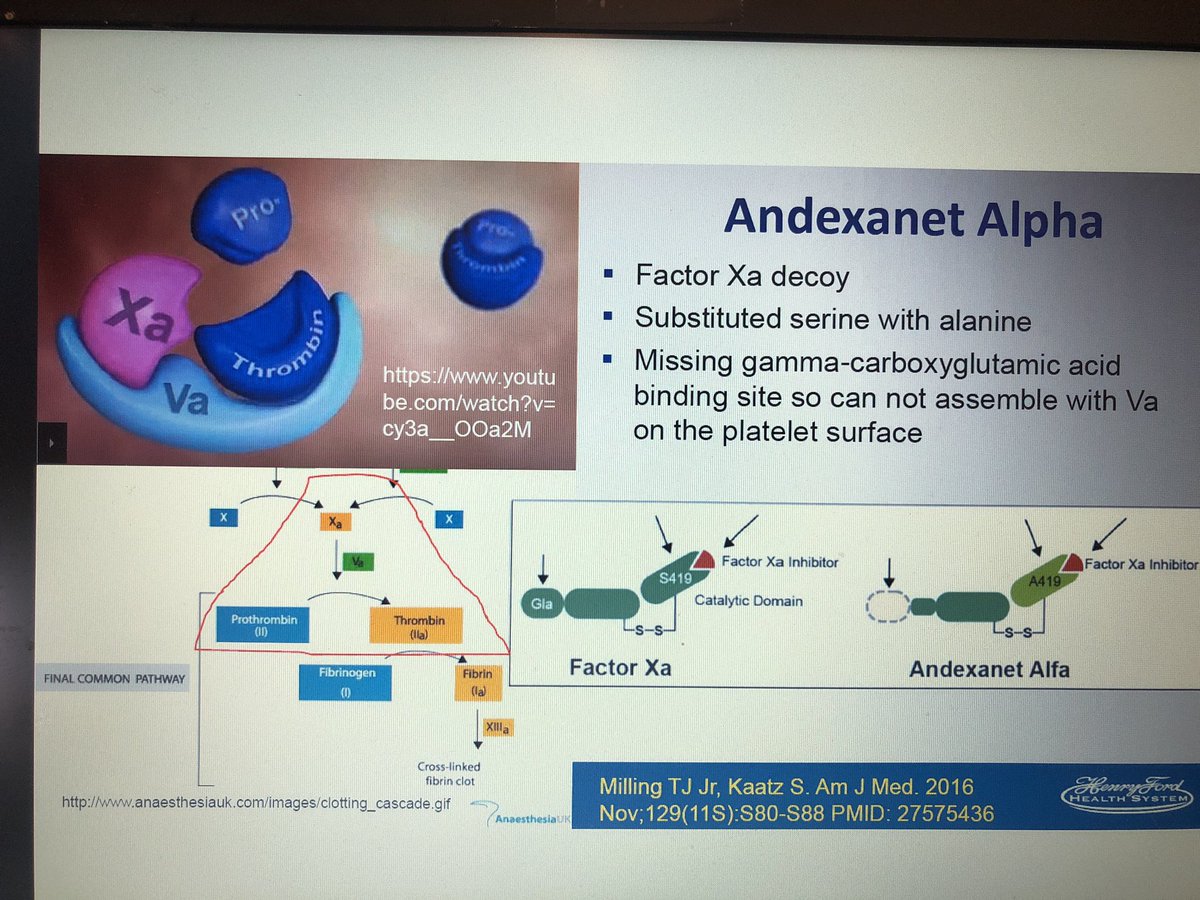
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
